Low-sampling antibody-antigen modelling tutorial using a local version of HADDOCK3
This tutorial consists of the following sections:
- Introduction
- HADDOCK general concepts
- A brief introduction to HADDOCK3
- Software and data setup
- Preparing PDB files for docking
- Defining restraints for docking
- Setting up and running the docking with HADDOCK3
- Analysis of docking results
- Conclusions
- BONUS 1: Does the antibody bind to a known interface of interleukin? ARCTIC-3D analysis
- BONUS 2: How good are AI-based models of antibody for docking?
- BONUS 3: Ensemble-docking using a combination of experimental and AI-predicted antibody structures
- BONUS 4: Antibody-antigen complex structure prediction from sequence using AlphaFold2
- Congratulations! 🎉
Introduction
This tutorial demonstrates the use of the new modular HADDOCK3 version for predicting the structure of an antibody-antigen complex using knowledge of the hypervariable loops on the antibody (i.e., the most basic knowledge) and epitope information identified from NMR experiments for the antigen to guide the docking.
An antibody is a large protein that generally works by attaching itself to an antigen, which is a unique site of the pathogen. The binding harnesses the immune system to directly attack and destroy the pathogen. Antibodies can be highly specific while showing low immunogenicity, which is achieved by their unique structure. The fragment crystallizable region (Fc region) activates the immune response and is species-specific, i.e. the human Fc region should not induce an immune response in humans. The fragment antigen-binding region (Fab region) needs to be highly variable to be able to bind to antigens of various nature (high specificity). In this tutorial we will concentrate on the terminal variable domain (Fv) of the Fab region.
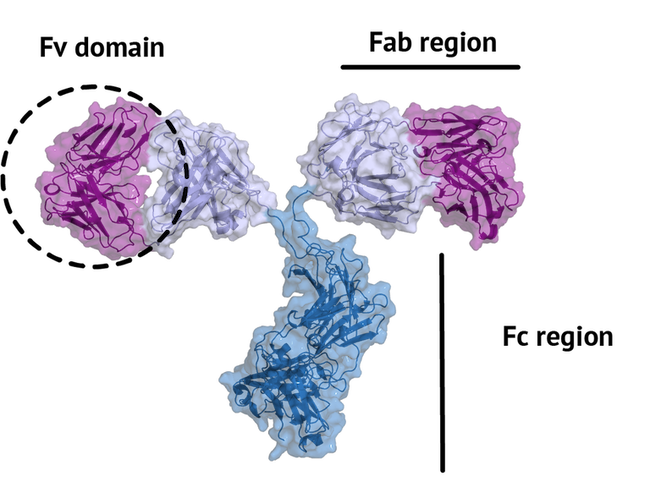
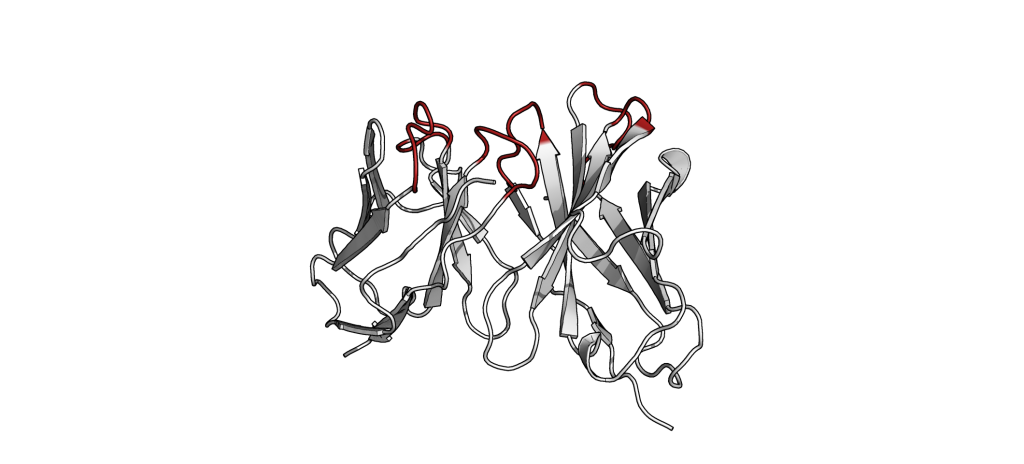
The small part of the Fab region that binds the antigen is called paratope. The part of the antigen that binds to an antibody is called epitope. The paratope consists of six highly flexible loops, known as complementarity-determining regions (CDRs) or hypervariable loops whose sequence and conformation are altered to bind to different antigens. CDRs are shown in red in the figure below:
In this tutorial we will be working with Interleukin-1β (IL-1β) (PDB code 4I1B) as an antigen and its highly specific monoclonal antibody gevokizumab (PDB code 4G6K) (PDB code of the complex 4G6M).
Throughout the tutorial, colored text will be used to refer to questions or instructions, and/or PyMOL commands.
This is a question prompt: try answering it! This an instruction prompt: follow it! This is a PyMOL prompt: write this in the PyMOL command line prompt! This is a Linux prompt: insert the commands in the terminal!
HADDOCK general concepts
HADDOCK (see https://www.bonvinlab.org/software/haddock2.4) is a collection of python scripts derived from ARIA (https://aria.pasteur.fr) that harness the power of CNS (Crystallography and NMR System – https://cns-online.org) for structure calculation of molecular complexes. What distinguishes HADDOCK from other docking software is its ability, inherited from CNS, to incorporate experimental data as restraints and use these to guide the docking process alongside traditional energetics and shape complementarity. Moreover, the intimate coupling with CNS endows HADDOCK with the ability to actually produce models of sufficient quality to be archived in the Protein Data Bank.
A central aspect to HADDOCK is the definition of Ambiguous Interaction Restraints or AIRs. These allow the translation of raw data such as NMR chemical shift perturbation or mutagenesis experiments into distance restraints that are incorporated in the energy function used in the calculations. AIRs are defined through a list of residues that fall under two categories: active and passive. Generally, active residues are those of central importance for the interaction, such as residues whose knockouts abolish the interaction or those where the chemical shift perturbation is higher. Throughout the simulation, these active residues are restrained to be part of the interface, if possible, otherwise incurring in a scoring penalty. Passive residues are those that contribute for the interaction, but are deemed of less importance. If such a residue does not belong in the interface there is no scoring penalty. Hence, a careful selection of which residues are active and which are passive is critical for the success of the docking.
A brief introduction to HADDOCK3
HADDOCK3 is the next generation integrative modelling software in the long-lasting HADDOCK project. It represents a complete rethinking and rewriting of the HADDOCK2.X series, implementing a new way to interact with HADDOCK and offering new features to users who can now define custom workflows.
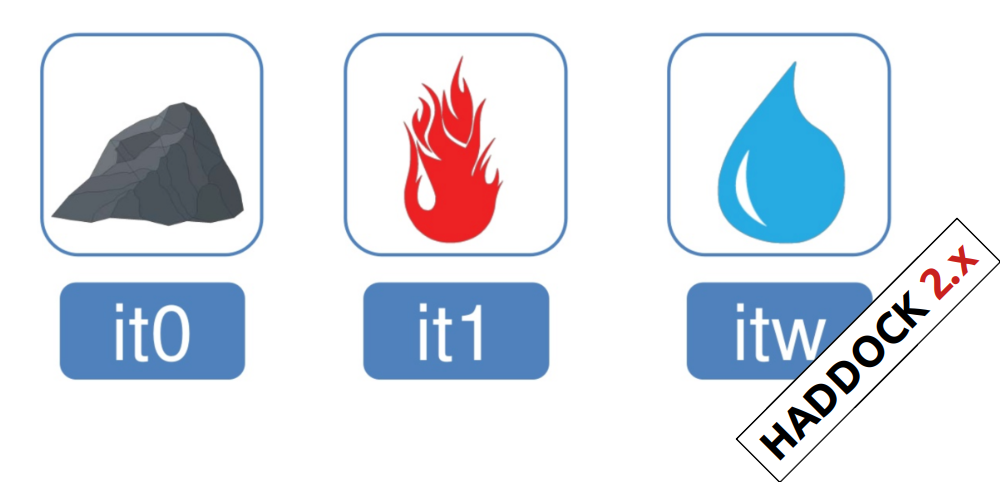
In the previous HADDOCK2.x versions, users had access to a highly
parameterisable yet rigid simulation pipeline composed of three steps:
rigid-body docking (it0), semi-flexible refinement (it1), and final refinement (itw).
In HADDOCK3, users have the freedom to configure docking workflows into
functional pipelines by combining the different HADDOCK3 modules, thus
adapting the workflows to their projects. HADDOCK3 has therefore developed to
truthfully work like a puzzle of many pieces (simulation modules) that users can
combine freely. To this end, the “old” HADDOCK machinery has been modularized,
and several new modules added, including third-party software additions. As a
result, the modularization achieved in HADDOCK3 allows users to duplicate steps
within one workflow (e.g., to repeat twice the it1 stage of the HADDOCK2.x
rigid workflow).
Note that, for simplification purposes, at this time, not all functionalities of HADDOCK2.x have been ported to HADDOCK3, which does not (yet) support NMR RDC, PCS and diffusion anisotropy restraints, cryo-EM restraints and coarse-graining. Any type of information that can be converted into ambiguous interaction restraints can, however, be used in HADDOCK3, which also supports the ab initio docking modes of HADDOCK.
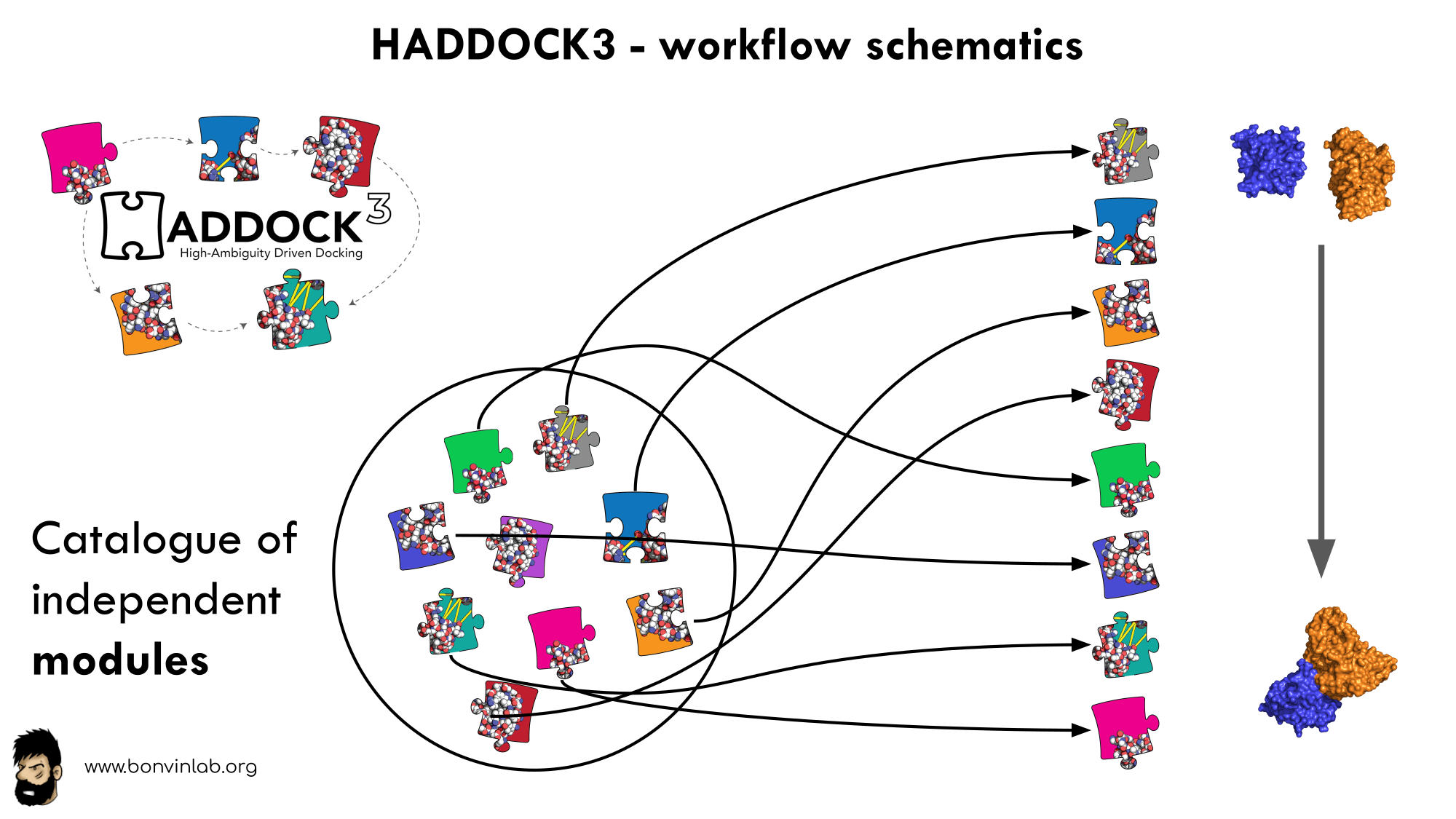
To keep HADDOCK3 modules organized, we catalogued them into several categories. But, there are no constraints on piping modules of different categories.
The main module categories are “topology”, “sampling”, “refinement”, “scoring”, and “analysis”. There is no limit to how many modules can belong to a category. Modules are added as developed, and new categories will be created if/when needed. You can access the HADDOCK3 documentation page for the list of all categories and modules. Below is a summary of the available modules:
- Topology modules
topoaa: generates the all-atom topologies for the CNS engine.
- Sampling modules
rigidbody: Rigid body energy minimization with CNS (it0in haddock2.x).lightdock: Third-party glow-worm swam optimization docking software.
- Model refinement modules
flexref: Semi-flexible refinement using a simulated annealing protocol through molecular dynamics simulations in torsion angle space (it1in haddock2.x).emref: Refinement by energy minimisation (itwEM only in haddock2.4).mdref: Refinement by a short molecular dynamics simulation in explicit solvent (itwin haddock2.X).
- Scoring modules
emscoring: scoring of a complex performing a short EM (builds the topology and all missing atoms).mdscoring: scoring of a complex performing a short MD in explicit solvent + EM (builds the topology and all missing atoms).
- Analysis modules
alascan: Performs a systematic (or user-define) alanine scanning mutagenesis of interface residues.caprieval: Calculates CAPRI metrics (i-RMSD, l-RMSD, Fnat, DockQ) with respect to the top scoring model or reference structure if provided.clustfcc: Clusters models based on the fraction of common contacts (FCC)clustrmsd: Clusters models based on pairwise RMSD matrix calculated with thermsdmatrixmodule.contactmap: Generate contact matrices of both intra- and intermolecular contacts and a chordchart of intermolecular contacts.rmsdmatrix: Calculates the pairwise RMSD matrix between all the models generated in the previous step.seletop: Selects the top N models from the previous step.seletopclusts: Selects top N clusters from the previous step.
The HADDOCK3 workflows are defined in simple configuration text files, similar to the TOML format but with extra features. Contrarily to HADDOCK2.X which follows a rigid (yet highly parameterisable) procedure, in HADDOCK3, you can create your own simulation workflows by combining a multitude of independent modules that perform specialized tasks.
Software and data setup
In order to follow this tutorial you will need to work on a Linux or MacOSX system. We will also make use of PyMOL (freely available for most operating systems) in order to visualize the input and output data. We will provide you links to download the various required software and data.
Further we are providing pre-processed PDB files for docking and analysis (but the preprocessing of those files will also be explained in this tutorial). The files have been processed to facilitate their use in HADDOCK and for allowing comparison with the known reference structure of the complex.
If you are running this tutorial on your own resources download and unzip the following zip archive and note the location of the extracted PDB files in your system. If running as part of the EU-ASEAN HPC school see the instructions below.
Note that you can also download and unzip this archive directly from the Linux command line:
Unziping the file will create the HADDOCK3-antibody-antigen directory which should contain the following directories and files:
docking-antibody-antigen-CDR-NMR-CSP.cfg: the HADDOCK3 configuration file used in this tutorialworkflows: a directory containing example haddock3 config files (workflows)pdbs: a directory ontains the pre-processed PDB filesrestraints: a directory containing the interface information and the corresponding restraint files for HADDOCKruns: a directory with pre-calculated resultsscripts: a directory containing various scripts used in this tutorial
EU-ASEAN 2023 HPC school
We will be making use of the Fugaku supercomputer for this tutorial. Please connect to fugaku using your credentials.
The software and data required for this tutorial have been pre-installed on Fugaku. In order to run the tutorial, first copy the required data into your home directory on fugagku:
unzip /vol0300/share/ra022304/LifeScience/20231213_Bonvin/HADDOCK3-antibody-antigen.zip
This will create the HADDOCK3-antibody-antigen directory with all necessary data and script and job examples ready for submission to the batch system.
HADDOCK3 has been pre-installed for the compute nodes. To test the installation, first create an interactive session on a node with:
pjsub --interact -L "node=1" -L "rscgrp=int" --sparam "wait-time=600" -L "elapse=01:00:00"
Once the session is active, activate HADDOCK3 with:
You can then test that haddock3 is indeed accessible with:
You should see a small help message explaining how to use the software.
View outputexpand_more
(haddock3)$ haddock3 -h
usage: haddock3 [-h] [--restart RESTART] [--extend-run EXTEND_RUN] [--setup]
[--log-level {DEBUG,INFO,WARNING,ERROR,CRITICAL}] [-v]
recipe
positional arguments:
recipe The input recipe file path
optional arguments:
-h, --help show this help message and exit
--restart RESTART Restart the run from a given step. Previous folders from the
selected step onward will be deleted.
--extend-run EXTEND_RUN
Start a run from a run directory previously prepared with the
`haddock3-copy` CLI. Provide the run directory created with
`haddock3-copy` CLI.
--setup Only setup the run, do not execute
--log-level {DEBUG,INFO,WARNING,ERROR,CRITICAL}
-v, --version show version
In case you want to obtain HADDOCK3 for another platform, navigate to its repository, fill the
registration form, and then follow the installation instructions.
Local setup (on your own)
If you are installing HADDOCK3 on your own system check the instructions are requirement below.
Installing HADDOCK3
To obtain HADDOCK3 navigate to its repository, fill the registration form, and then follow the installation instructions.
Installing CNS
The other required piece of software to run HADDOCK is its computational engine,
CNS (Crystallography and NMR System –
https://cns-online.org). CNS is
freely available for non-profit organizations. In order to get access to all
features of HADDOCK you will need to compile CNS using the additional files
provided in the HADDOCK distribution in the extras/cns1.3 directory. Compilation of
CNS might be non-trivial. Some guidance on installing CNS is provided in the online
HADDOCK3 documentation page here.
Once CNS has been properly compiled, you will have to copy the executable to haddock3/bin/cns and make sure it is executable and work. Try starting cns from the command line. You should see the following output:
View CNS prompt outputexpand_more
============================================================
| |
| Crystallography & NMR System (CNS) |
| CNSsolve |
| |
============================================================
Version: 1.3 at patch level U
Status: Special UU release with Rg, paramagnetic
and Z-restraints (A. Bonvin, UU 2013)
============================================================
Written by: A.T.Brunger, P.D.Adams, G.M.Clore, W.L.DeLano,
P.Gros, R.W.Grosse-Kunstleve,J.-S.Jiang,J.M.Krahn,
J.Kuszewski, M.Nilges, N.S.Pannu, R.J.Read,
L.M.Rice, G.F.Schroeder, T.Simonson, G.L.Warren.
Copyright (c) 1997-2010 Yale University
============================================================
Running on machine: hostname unknown (Linux,64-bit)
Program started by: l00902
Program started at: 16:34:22 on 06-Dec-2023
============================================================
FFT3C: Using FFTPACK4.1
CNSsolve>
Exit the CNS command line by typing stop.
Auxiliary software
PDB-tools: A useful collection of Python scripts for the
manipulation (renumbering, changing chain and segIDs…) of PDB files is freely
available from our GitHub repository. pdb-tools is automatically installed
with HADDOCK3. If you have activated the HADDOCK3 Python environment you have
access to the pdb-tools package.
PyMol: In this tutorial we will make use of PyMol for visualization. If not already installed on your system, download and install PyMol. Note that you can use your favorite visulation software but instructions are only provided here for PyMol.
Preparing PDB files for docking
In this section we will prepare the PDB files of the antibody and antigen for docking. Crystal structures of both the antibody and the antigen in their free forms are available from the PDBe database.
Important: For a docking run with HADDOCK, each molecule should consist of a single chain with non-overlapping residue numbering.
As an antibody consists of two chains (L+H), we will have to prepare it for use in HADDOCK. For this we will be making use of pdb-tools from the command line.
Note that pdb-tools is also available as a web service.
Preparing the antibody structure
Using PDB-tools we will download the unbound structure of the antibody from the PDB database (the PDB ID is 4G6K) and then process it to have a unique chain ID (A) and non-overlapping residue numbering by renumbering the merged pdb (starting from 1).
This can be done from the command line with:
pdb_fetch 4G6K | pdb_tidy -strict | pdb_selchain -H | pdb_delhetatm | pdb_fixinsert | pdb_keepcoord | pdb_selres -1:120 | pdb_tidy -strict > 4G6K_H.pdb pdb_fetch 4G6K | pdb_tidy -strict | pdb_selchain -L | pdb_delhetatm | pdb_fixinsert | pdb_keepcoord | pdb_selres -1:107 | pdb_tidy -strict > 4G6K_L.pdb pdb_merge 4G6K_H.pdb 4G6K_L.pdb | pdb_reres -1 | pdb_chain -A | pdb_chainxseg | pdb_reres -1 | pdb_tidy -strict > 4G6K_clean.pdb
The first command fetches the PDB ID, selects the heavy chain (H) (pdb_selchain) and removes water and heteroatoms (pdb_delhetatm) (in this case no co-factor is present that should be kept).
An important part for antibodies is the pdb_fixinsert command that fixes the residue numbering of the HV loops: Antibodies often follow the Chothia numbering scheme and insertions created by this numbering scheme (e.g. 82A, 82B, 82C) cannot be processed by HADDOCK directly (if not done those residues will not be considered resulting effectively in a break in the loop). As such renumbering is necessary before starting the docking.
Then, the command pdb_selres selects only the residues from 1 to 120, so as to consider only the variable domain (FV) of the antibody. This allows to save a substantial amount of computational resources.
The second command does the same for the light chain (L) with the difference that the light chain is slightly shorter and we can focus on the first 107 residues.
The third and last command merges the two processed chains, renumber the residues starting from 1 (pdb_reres) and assign them unique chain and segIDs (pdb_chain and pdb_chainxseg), resulting in the HADDOCK-ready 4G6K_clean.pdb file. You can view its sequence running:
Note The ready-to-use file can be found in the pdbs directory of the provided tutorial data.
Preparing the antigen structure
Using PDB-tools we will download the unbound structure of Interleukin-1β from the PDB database (the PDB ID is 4I1B), remove the hetero atoms and then process it to assign it chainID B.
Important: Each molecule given to HADDOCK in a docking scenario must have a unique chainID/segID.
Defining restraints for docking
Before setting up the docking we need first to generate distance restraint files in a format suitable for HADDOCK. HADDOCK uses CNS as computational engine. A description of the format for the various restraint types supported by HADDOCK can be found in our Nature Protocol paper, Box 4.
Distance restraints are defined as follows:
assi (selection1) (selection2) distance, lower-bound correction, upper-bound correction
The lower limit for the distance is calculated as: distance minus lower-bound correction and the upper limit as: distance plus upper-bound correction
The syntax for the selections can combine information about:
- chainID -
segidkeyword - residue number -
residkeyword - atom name -
namekeyword.
Other keywords can be used in various combinations of OR and AND statements. Please refer for that to the online CNS manual.
E. g. a distance restraint between the CB carbons of residues 10 and 200 in chains A and B with an allowed distance range between 10 and 20Å would be defined as follows:
assi (segid A and resid 10 and name CB) (segid B and resid 200 and name CB) 20.0 10.0 0.0
Identifying the paratope of the antibody
Nowadays there are several computational tools that can identify the paratope (the residues of the hypervariable loops involved in binding) from the provided antibody sequence. In this tutorial we are providing you the corresponding list of residue obtained using ProABC-2. ProABC-2 uses a convolutional neural network to identify not only residues which are located in the paratope region but also the nature of interactions they are most likely involved in (hydrophobic or hydrophilic). The work is described in Ambrosetti, et al Bioinformatics, 2020.
The corresponding paratope residues (those with either an overall probability >= 0.4 or a probability for hydrophobic or hydrophilic > 0.3) are:
31,32,33,34,35,52,54,55,56,100,101,102,103,104,105,106,151,152,169,170,173,211,212,213,214,216
The numbering corresponds to the numbering of the 4G6K_clean.pdb PDB file.
Let us visualize those onto the 3D structure.
For this start PyMOL and load 4G6K_clean.pdb
File menu -> Open -> select 4G6K_clean.pdb
Alternatively, if PyMol is accessible from the command line simply type:
We will now highlight the predicted paratope. In PyMOL type the following commands:
color white, all
select paratope, (resi 31+32+33+34+35+52+54+55+56+100+101+102+103+104+105+106+151+152+169+170+173+211+212+213+214+216)
color red, paratope
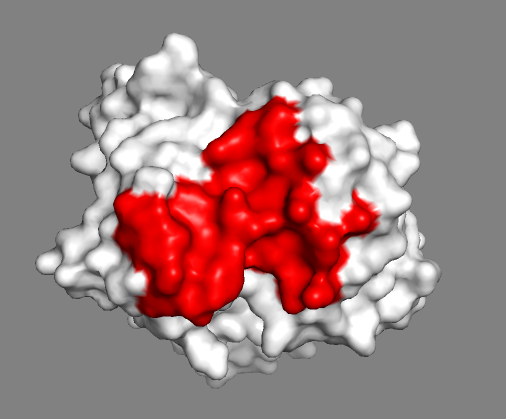
Let us now switch to a surface representation to inspect the predicted binding site.
Inspect the surface.
Do the identified paratope residues form a well defined patch on the surface?
See surface view of the paratope expand_more
Identifying the epitope of the antigen
The article describing the crystal structure of the antibody-antigen complex we are modelling also reports on experimental NMR chemical shift titration experiments to map the binding site of the antibody (gevokizumab) on Interleukin-1β. The residues affected by binding are listed in Table 5 of Blech et al. JMB 2013:
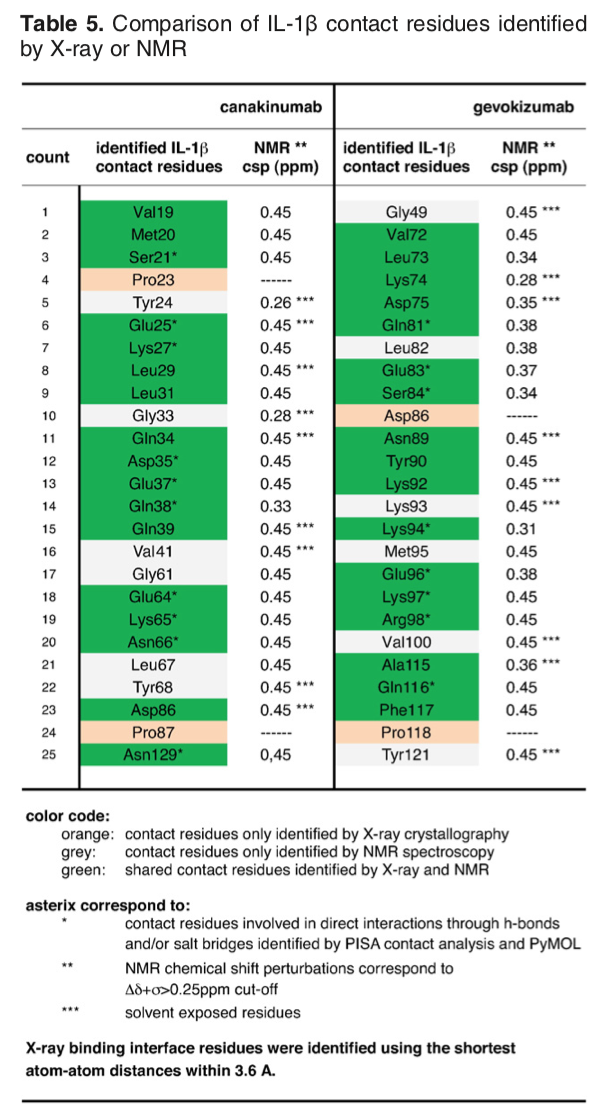
The list of binding site (epitope) residues identified by NMR is:
72,73,74,75,81,83,84,89,90,92,94,96,97,98,115,116,117
We will now visualize the epitope on Interleukin-1β. For this start PyMOL and from the PyMOL File menu open the provided PDB file of the antigen.
File menu -> Open -> select 4I1B_clean.pdb
color white, all show surface select epitope, (resi 72+73+74+75+81+83+84+89+90+92+94+96+97+98+115+116+117) color red, epitope
Inspect the surface.
Do the identified residues form a well defined patch on the surface?
The answer to that question should be yes, but we can see some residues not colored that might also be involved in the binding - there are some white spots around/in the red surface.
See surface view of the epitope identified by NMR expand_more

In HADDOCK we are dealing with potentially incomplete binding sites by defining surface neighbors as passive residues. These are added to the definition of the interface but will not lead to any energetic penalty if they are not part of the binding site in the final models, while the residues defined as active (typically the identified or predicted binding site residues) will. When using the HADDOCK server, passive residues will be automatically defined. Here since we are
using a local version, we need to define those manually.
This can easily be done using a haddock3 command line tool in the following way:
The command returns a list of passive residues which you should save to a file for further use.
We can visualize the epitope and its surface neighbors using PyMOL:
File menu -> Open -> select 4I1B_clean.pdb
color white, all show surface select epitope, (resi 72+73+74+75+81+83+84+89+90+92+94+96+97+98+115+116+117) color red, epitope select passive, (resi 3+24+46+47+48+50+66+76+77+79+80+82+86+87+88+91+93+95+118+119+120) color green, passive
See the epitope and passive residues expand_more

The NMR-identified residues and their surface neighbors generated with the above command can be used to define ambiguous interactions restraints, either using the NMR identified residues as active in HADDOCK, or combining those with the surface neighbors.
The difference between active and passive residues in HADDOCK is:
active residues will be “forced” to be at the interface, meaning that if the end models they are not an energetic penalty will be paid. The interface in this context is defined by the union of active and passive residues on the partner molecules.
passive residues should be at the interface, if they are not no energetic penalty is paid.
In general it is better to be too generous rather than too strict in the definition of passive residues. An important aspect is to filter both the active (the residues identified from
your mapping experiment) and passive residues by their solvent accessibility. This is done automatically when using the haddock3-restraints passive_from_active command: residues with less that 15% relative solvent accessibility (same cutoff as the default in the HADDOCK server) are discarded.
This is however not a hard limit and you might consider including even more buried residues if some
important chemical group seems solvent accessible from a visual inspection.
Defining ambiguous restraints
Once you have identified your active and passive residues for both molecules, you can proceed with the generation of the ambiguous interaction restraints (AIR) file for HADDOCK. For this you can either make use of our online GenTBL web service, entering the list of active and passive residues for each molecule, the chainIDs of each molecule and saving the resulting restraint list to a text file, or use again the haddock3-restraints command.
To use our haddock3-restraints active_passive_to_ambig script you need to
create for each molecule a file containing two lines:
- The first line corresponds to the list of active residues (numbers separated by spaces)
- The second line corresponds to the list of passive residues (numbers separated by spaces).
Important: The file must consist of two lines, but a line could be empty (e.g. if you do not want to define active residues for one molecule). There must however be at least one set of active residue defined for one of the molecule.
- For the antibody we will use the predicted paratope as active and no passive residues defined. The corresponding file can be found in the
restraintsdirectory asantibody-paratope.act-pass:
1 32 33 34 35 52 54 55 56 100 101 102 103 104 105 106 151 152 169 170 173 211 212 213 214 216
- For the antigen we will use the NMR-identified epitope as active and the surface neighbors as passive. The corresponding file can be found in the
restraintsdirectory asantigen-NMR-epitope.act-pass:
72 73 74 75 81 83 84 89 90 92 94 96 97 98 115 116 117 3 24 46 47 48 50 66 76 77 79 80 82 86 87 88 91 93 95 118 119 120
Using those two files, we can generate the CNS-formatted AIR restraint files with the following command:
This generates a file called ambig-paratope-NMR-epitope.tbl that contains the AIR
restraints.
Inspect the generated file and note how the ambiguous distances are defined.
View an extract of the AIR file expand_more
assign (resi 31 and segid A)
(
(resi 72 and segid B)
or
(resi 73 and segid B)
or
(resi 74 and segid B)
or
(resi 75 and segid B)
or
(resi 81 and segid B)
or
(resi 83 and segid B)
or
(resi 84 and segid B)
or
(resi 89 and segid B)
or
(resi 90 and segid B)
or
(resi 92 and segid B)
or
(resi 94 and segid B)
or
(resi 96 and segid B)
or
(resi 97 and segid B)
or
(resi 98 and segid B)
or
(resi 115 and segid B)
or
(resi 116 and segid B)
or
(resi 117 and segid B)
or
(resi 3 and segid B)
or
(resi 24 and segid B)
or
(resi 46 and segid B)
or
(resi 47 and segid B)
or
(resi 48 and segid B)
or
(resi 50 and segid B)
or
(resi 66 and segid B)
or
(resi 76 and segid B)
or
(resi 77 and segid B)
or
(resi 79 and segid B)
or
(resi 80 and segid B)
or
(resi 82 and segid B)
or
(resi 86 and segid B)
or
(resi 87 and segid B)
or
(resi 88 and segid B)
or
(resi 91 and segid B)
or
(resi 93 and segid B)
or
(resi 95 and segid B)
or
(resi 118 and segid B)
or
(resi 119 and segid B)
or
(resi 120 and segid B)
) 2.0 2.0 0.0
...
See answer expand_more
The default distance range for those is between 0 and 2Å, which might seem short but makes senses because of the 1/r^6 summation in the AIR energy function that makes the effective distance to be significantly shorter than the shortest distance entering the sum.The effective distance is calculated as the SUM over all pairwise atom-atom distance combinations between an active residue and all the active+passive on the other molecule: SUM[1/r^6]^(-1/6).
Restraints validation
If you modify manually this generated restraint files or create your own, it is possible to quickly check if the format is valid using the following haddock3 command:
haddock3-restraints validate_tbl ambig-paratope-NMR-epitope.tbl --silent
No output means that your TBL file is valid.
Note that this only validates the syntax of the restraint file, but does not check if the selections defined in the restraints are actually existing in your input PDB files.
Additional restraints for multi-chain proteins
As an antibody consists of two separate chains, it is important to define a few distance restraints
to keep them together during the high temperature flexible refinement stage of HADDOCK otherwise they might slightly drift appart. This can easily be done using the haddock3-restraints restrain_bodies subcommand.
haddock3-restraints restrain_bodies 4G6K_clean.pdb > antibody-unambig.tbl
The result file contains two CA-CA distance restraints with the exact distance measured between two randomly picked CA atoms:
assign (segid A and resi 110 and name CA) (segid A and resi 132 and name CA) 47.578 0.0 0.0 assign (segid A and resi 97 and name CA) (segid A and resi 204 and name CA) 33.405 0.0 0.0
This file is also provided in the restraints directory.
Setting up and running the docking with HADDOCK3
Now that we have all required files at hand (PDB and restraints files) it is time to setup our docking protocol. In this tutorial, considering we have rather good information about the paratope and epitope, we will execute a fast HADDOCK3 docking workflow, reducing the non-negligible computational cost of HADDOCK by decreasing the sampling, without impacting too much the accuracy of the resulting models.
HADDOCK3 workflow definition
The first step is to create a HADDOCK3 configuration file that will define the docking workflow. We will follow a classic HADDOCK workflow consisting of rigid body docking, semi-flexible refinement and final energy minimisation followed by clustering.
We will also integrate various analysis modules in our workflow:
-
caprievalwill be used at various stages to compare models to either the best scoring model (if no reference is given) or a reference structure, which in our case we have at hand. This will directly allow us to assess the performance of the protocol. In the absence of a reference,caprievalis still usefull to assess the convergence of a run and analyse the results. -
contactmapadded as last module will generate contact matrices of both intra- and intermolecular contacts and a chordchart of intermolecular contacts for each cluster.
Our workflow consists of the following modules:
topoaa: Generates the topologies for the CNS engine and build missing atomsrigidbody: Rigid body energy minimisation (it0in haddock2.x)caprieval: Calculates CAPRI metrics (i-RMSD, l-RMSD, Fnat, DockQ) with respect to the top scoring model or reference structure if providedseletop: Selects the top X models from the previous moduleflexref: Semi-flexible refinement of the interface (it1in haddock2.4)caprievalemref: Final refinement by energy minimisation (itwEM only in haddock2.4)caprievalclustfcc: Clustering of models based on the fraction of common contacts (FCC)seletopclusts: Selects the top models of all clusterscaprievalcontactmap: Contacts matrix and a chordchart of intermolecular contacts
The corresponding toml configuration file (provided in workflows/docking-antibody-antigen-CDR-NMR-CSP.cfg) looks like:
# ====================================================================
# Antibody-antigen docking example with restraints from the antibody
# paratope to the NMR-identified epitope on the antigen
# ====================================================================
# directory in which the scoring will be done
run_dir = "run1-CDR-NMR-CSP"
# compute mode
mode = "local"
ncores=50
# Self contained rundir (to avoid problems with long filename paths)
self_contained = true
molecules = [
"pdbs/4G6K_clean.pdb",
"pdbs/4I1B_clean.pdb"
]
# ====================================================================
# Parameters for each stage are defined below, prefer full paths
# ====================================================================
[topoaa]
[rigidbody]
# CDR to NMR epitope ambig restraints
ambig_fname = "restraints/ambig-paratope-NMR-epitope.tbl"
# Restraints to keep the antibody chains together
unambig_fname = "restraints/antibody-unambig.tbl"
sampling = 50
[caprieval]
reference_fname = "pdbs/4G6M_matched.pdb"
[seletop]
select = 50
[flexref]
tolerance = 5
# CDR to NMR epitope ambig restraints
ambig_fname = "restraints/ambig-paratope-NMR-epitope.tbl"
# Restraints to keep the antibody chains together
unambig_fname = "restraints/antibody-unambig.tbl"
[caprieval]
reference_fname = "pdbs/4G6M_matched.pdb"
[emref]
tolerance = 5
# CDR to NMR epitope ambig restraints
ambig_fname = "restraints/ambig-paratope-NMR-epitope.tbl"
# Restraints to keep the antibody chains together
unambig_fname = "restraints/antibody-unambig.tbl"
[caprieval]
reference_fname = "pdbs/4G6M_matched.pdb"
[clustfcc]
[seletopclusts]
top_models = 4
[caprieval]
reference_fname = "pdbs/4G6M_matched.pdb"
[contactmap]
# ====================================================================In this case since we have information for both interfaces we use a low-sampling configuration file, which takes only a small amount of computational resources to run. From the sampling parameters in the above config file you can see we are sompling 50 models at each stage of the docking:
The initial sampling parameter at the rigid-body energy minimization (rigidbody) module is set to 50 models, all of which are passed to the flexible refinement (flexref) module with the seletop module. The subsequence flexible refinement (flexref module) and energy minimisation (emref) modules will use all models passed by the seletop module. FCC clustering (clustfcc) is then applied to group together models sharing a consistent fraction of the interface contacts. The top 4 models of each cluster are saved to disk (seletopclusts). Multiple caprieval modules are executed at different stages of the workflow to check how the quality (and rankings) of the models change throughout the protocol.
To get a list of all possible parameters that can be defined in a specific module (and their default values) you can use the following command:
Add the -d option to get a more detailed description of parameters and use the -h option to see a list of arguments and options.
Note that, in contrast to HADDOCK2.X, we have much more flexibility in defining our workflow. As an example, we could use this flexibility by introducing a clustering step after the initial rigid-body docking stage, select a given number of models per cluster and refine all of those. For an example of this strategy see the BONUS 3 section about ensemble docking.
Running HADDOCK3
In in the first section of the workflow above we have a parameter mode defining the execution mode. HADDOCK3 currently supports three difference execution modes:
- local : In this mode HADDOCK3 will run on the current system, using the defined number of cores (
ncores) in the config file to a maximum of the total number of available cores on the system - batch: in this mode HADDOCK3 will typically be started on your local server (e.g. the login node) and will dispatch jobs to the batch system of your cluster (slurm and torque are currently supported);
- mpi: HADDOCK3 supports a pseudo parallel MPI implementation which allows to harvest the power of mutliple nodes to distribute the computations (functional but still very experimental at this stage).
Execution of Fugaku using a full node (EU-ASEAN HPC School)
To execute the workflow on Fugaku, we will create a job file that will execute HADDOCK3 on a node, with HADDOCK3 running in local mode (the setup in the above configuration file with mode="local") and harvesting all core of that node (ncores=50).
Here is an example of such an execution script (also provided in the workflows directory as run-haddock3-fugaku.sh):
#!/bin/sh
#PJM -g ra022304
#PJM -L "rscgrp=small"
#PJM -L "node=1"
#PJM -L "elapse=01:00:00"
#PJM -x PJM_LLIO_GFSCACHE=/vol0004:/vol0003
#PJM -s # Statistical information output
source /vol0300/share/ra022304/LifeScience/20231213_Bonvin/miniconda3/etc/profile.d/conda.sh
conda activate haddock3
haddock3 docking-antibody-antigen-CDR-NMR-CSP.cfgThis file should be submitted to the batch system using the pjsub command:
pjsub workflows/run-haddock3-fugaku.sh
This run should take about 20 minutes to complete on a single node using 50 arm cores.
Local execution
In this mode HADDOCK3 will run on the current system, using the defined number of cores (ncores) in the config file to a maximum of the total number of available cores on the system minus one. An example of the relevant parameters to be defined in the first section of the config file is:
# compute mode
mode = "local"
# 1 nodes x 50 ncores
ncores = 50In this mode HADDOCK3 can be started from the command line with as argument the configuration file of the defined workflow.
haddock3 <my-workflow-configuration-file>
Alternatively redirect the output to a log file and send haddock3 to the background.
As an indication, running locally on an Apple M2 laptop using 10 cores, this workflow completed in 7 minutes.
haddock3 <my-workflow-configuration-file> > haddock3.log &
Note: This is also the execution mode that should be used for example when submitting the HADDOCK3 job to a node of a cluster, requesting X number of cores.
View an example script for submitting via the slurm batch system expand_more
#!/bin/bash
#SBATCH --nodes=1
#SBATCH --tasks-per-node=50
#SBATCH -J haddock3
#SBATCH --partition=medium
# activate the haddock3 conda environment
source $HOME/miniconda3/etc/profile.d/conda.sh
conda activate haddock3
# go to the run directory
cd $HOME/HADDOCK3-antibody-antigen
# execute
haddock3 \<my-workflow-configuration-file\>
Exection in batch mode using slurm
In this mode HADDOCK3 will typically be started on your local server (e.g. the login node) and will dispatch jobs to the batch system of your cluster. Two batch systems are currently supported: slurm and torque (defined by the batch_type parameter). In the configuration file you will
have to define the queue name and the maximum number of concurrent jobs sent to the queue (queue_limit).
Since HADDOCK3 single model calculations are quite fast, it is recommended to calculate multiple models within one job submitted to the batch system. The number of model per job is defined by the concat parameter in the configuration file. You want to avoid sending thousands of very short jobs to the batch system if you want to remain friend with your system administrators…
An example of the relevant parameters to be defined in the first section of the config file is:
# compute mode
mode = "batch"
# batch system
batch_type = "slurm"
# queue name
queue = "short"
# number of concurrent jobs to submit to the batch system
queue_limit = 100
# number of models to produce per submitted job
concat = 10In this mode HADDOCK3 can be started from the command line as for the local mode.
MPI mode
HADDOCK3 supports a parallel pseudo-MPI implementation (functional but still very experimental at this stage). For this to work, the mpi4py library must have been installed at installation time. Refer to the MPI-related instructions.
The execution mode should be set to mpi and the total number of cores should match the requested resources when submitting to the batch system.
An example of the relevant parameters to be defined in the first section of the config file is:
# compute mode
mode = "mpi"
# 5 nodes x 50 tasks = ncores = 250
ncores = 250In this execution mode the HADDOCK3 job should be submitted to the batch system requesting the corresponding number of nodes and cores per node.
View an example script for submitting an MPI HADDOCK3 job the slurm batch system expand_more
#!/bin/bash
#SBATCH --nodes=5
#SBATCH --tasks-per-node=50
#SBATCH -J haddock3mpi
# Make sure haddock3 is activated
source $HOME/miniconda3/etc/profile.d/conda.sh
conda activate haddock3
# go to the run directory
# edit if needed to specify the correct location
cd $HOME/HADDOCK3-antibody-antigen
# execute
haddock3 \<my-workflow-configuration-file\>
Analysis of docking results
In case something went wrong with the docking (or simply if you do not want to wait for the results) you can find the following precalculated runs in the runs directory:
run1: run started using the unbound antibodyrun1-af2: run started using the Alphafold-multimer antibody (see BONUS 2)run1-abb: run started using the Immunebuilder antibody (see BONUS 2)run1-ens: run started using an ensemble of antibody models (see BONUS 3)
Once your run has completed inspect the content of the resulting directory. You will find the various steps (modules) of the defined workflow numbered sequentially startin at 0, e.g.:
> ls run1/
00_topoaa/
01_rigidbody/
02_caprieval/
03_seletop/
04_flexref/
05_caprieval/
06_emref/
07_caprieval/
08_clustfcc/
09_seletopclusts/
10_caprieval/
11_contactmap/
analysis/
data/
log
toppar/
traceback/There is in addition the log file (text file) and four additional directories:
- the
analysisdirectory containing various plots to visualise the results for eachcaprievalstep and a general report (report.html) that provides all statistics with various plot. You can open this file in your preferred web browser. - the
datadirectory containing the input data (PDB and restraint files) for the various modules - the
toppardirectory containing the force field topology and paramter files (only present when running in self-contained mode) - the
tracebackdirectory containingtraceback.tsvthat links all models in order to see which model originates from which throughout all steps of the workflow You can find information about the duration of the run at the bottom of the log file. Each sampling/refinement/selection module will contain PDB files.
For example, the 09_seletopclusts directory contains the selected models from each cluster. The clusters in that directory are numbered based
on their rank, i.e. cluster_1 refers to the top-ranked cluster. Information about the origin of these files can be found in that directory in the seletopclusts.txt file.
The simplest way to extract ranking information and the corresponding HADDOCK scores is to look at the 10_caprieval directories (which is why it is a good idea to have it as the final module, and possibly as intermediate steps). This directory will always contain a capri_ss.tsv single model statistics file, which contains the model names, rankings and statistics (score, iRMSD, Fnat, lRMSD, ilRMSD and dockq score). E.g.:
model md5 caprieval_rank score irmsd fnat lrmsd ilrmsd dockq cluster-id cluster-ranking model-cluster-ranking air angles bonds bsa cdih coup dani desolv dihe elec improper rdcs rg sym total vdw vean xpcs ../06_emref/emref_4.pdb - 1 -145.275 1.034 0.879 2.856 1.929 0.819 - - - 69.699 0.000 0.000 1976.860 0.000 0.000 0.000 6.775 0.000 -493.142 0.000 0.000 0.000 0.000 -483.835 -60.391 0.000 0.000 ../06_emref/emref_6.pdb - 2 -132.350 1.255 0.828 2.269 2.166 0.783 - - - 67.962 0.000 0.000 1882.240 0.000 0.000 0.000 11.799 0.000 -510.162 0.000 0.000 0.000 0.000 -491.112 -48.912 0.000 0.000 ../06_emref/emref_1.pdb - 3 -131.650 0.822 0.862 2.060 1.269 0.858 - - - 79.712 0.000 0.000 1879.870 0.000 0.000 0.000 -2.596 0.000 -487.314 0.000 0.000 0.000 0.000 -447.165 -39.562 0.000 0.000 ../06_emref/emref_2.pdb - 4 -129.551 1.296 0.776 2.308 2.364 0.760 - - - 129.685 0.000 0.000 1862.120 0.000 0.000 0.000 1.240 0.000 -508.711 0.000 0.000 0.000 0.000 -421.043 -42.018 0.000 0.000 ...
If clustering was performed prior to calling the caprieval module the capri_ss.tsv file will also contain information about to which cluster the model belongs to and its ranking within the cluster.
The relevant statistics are:
- score: the HADDOCK score (arbitrary units)
- irmsd: the interface RMSD, calculated over the interfaces the molecules
- fnat: the fraction of native contacts
- lrmsd: the ligand RMSD, calculated on the ligand after fitting on the receptor (1st component)
- ilrmsd: the interface-ligand RMSD, calculated over the interface of the ligand after fitting on the interface of the receptor (more relevant for small ligands for example)
- dockq: the DockQ score, which is a combination of irmsd, lrmsd and fnat and provides a continuous scale between 1 (exactly equal to reference) and 0
Various other terms are also reported including:
- bsa: the buried surface area in squared Angstromn
- elec: the intermolecular electrostatic energy
- vdw: the intermolecular van der Waals energy
- desolv: the desolvation energy
The iRMSD, lRMSD and Fnat metrics are the ones used in the blind protein-protein prediction experiment CAPRI (Critical PRediction of Interactions).
In CAPRI the quality of a model is defined as (for protein-protein complexes):
- acceptable model: i-RMSD < 4Å or l-RMSD<10Å and Fnat > 0.1 (0.23 < DOCKQ < 0.49)
- medium quality model: i-RMSD < 2Å or l-RMSD<5Å and Fnat > 0.3 (0.49 < DOCKQ < 0.8)
- high quality model: i-RMSD < 1Å or l-RMSD<1Å and Fnat > 0.5 (DOCKQ > 0.8)
What is based on this CAPRI criterion the quality of the best model listed above (emref_6.pdb)?
In case the caprieval module is called after a clustering step an additional file will be present in the directory: capri_clt.tsv.
This file contains the cluster ranking and score statistics, averaged over the minimum number of models defined for clustering
(4 by default), with their corresponding standard deviations. E.g.:
cluster_rank cluster_id n under_eval score score_std irmsd irmsd_std fnat fnat_std lrmsd lrmsd_std dockq dockq_std air air_std bsa bsa_std desolv desolv_std elec elec_std total total_std vdw vdw_std caprieval_rank 1 1 13 - -134.706 6.188 1.102 0.190 0.836 0.039 2.373 0.294 0.805 0.037 86.764 25.183 1900.272 44.896 4.305 5.461 -499.832 9.836 -460.789 28.355 -47.721 8.079 1 2 2 10 - -103.801 6.412 5.072 0.072 0.125 0.007 11.718 0.592 0.184 0.008 119.323 25.301 1602.258 76.499 8.380 2.269 -300.700 36.597 -245.350 36.102 -63.973 3.596 2 3 3 9 - -96.625 5.281 9.931 0.292 0.069 0.012 19.389 0.457 0.084 0.005 163.724 58.987 1525.055 31.387 2.803 1.489 -367.539 27.148 -246.109 63.575 -42.293 5.960 3 4 4 4 - -96.089 15.346 14.725 0.026 0.090 0.007 23.152 0.311 0.073 0.003 174.066 46.134 1793.750 120.512 3.950 1.692 -332.615 23.798 -209.472 70.181 -50.923 6.901 4
In this file you find the cluster rank, the cluster ID (which is related to the size of the cluster, 1 being always the largest cluster), the number of models (n) in the cluster and the corresponding statistics (averages + standard deviations). The corresponding cluster PDB files will be found in the processing 09_seletopclusts directory.
While these simple text file can be easily checked from the command line already, they might be cumbersome to read. For that reason we have developed a post-processing analysis that automatically generates html reports for all caprieval steps in the workflow. These are located in the respective analysis/XX_caprieval directories and can be viewed using your favorite web browser.
Cluster statistics
Let us now analyse the docking results. Use for that either your own run or a pre-calculated run provided in the runs directory.
Go into the analysis/10_caprieval_analysis directory of the respective run directory (if needed copy the run or that directory to your local computer) and open in a web browser the report.html file. Be patient as this page contains interactive plots that take some time to generate.
On the top of the page you will see a table that summarises the cluster statistics (taken from the capri_clt.tsv file).
The columns (corresponding to the various clusters) are sorted by default on the cluster rank, which is based on the HADDOCK score (found on row 4 of the table).
As this is an interactive table you can sort it as you wish by using the arrows present in the first column. Simply click on the arrows of the term you want to use to sort the table (and you can sort it in ascending or descending order). A snapshot of this table is shown below.

You can also view this report online here
Note that in case the PDB files are still compressed (gzipped) the download links will not work. Also online visualisation is not enabled.
Inspect the final cluster statistics
How many clusters are generated?
Since for this tutorial we have at hand the crystal structure of the complex, we provided it as reference to the caprieval modules.
This means that the iRMSD, lRMSD, Fnat and DockQ statistics report on the quality of the docked model compared to the reference crystal structure.
How many clusters of acceptable or better quality have been generate according to CAPRI criteria?
What is the rank of the best cluster generated?
What is the rank of the first acceptable of better cluster generated?
Visualizing the scores and their components
Next to the cluster statistic table shown above, the report.html file also contains a variety of plots plotting the HADDOCK score
and its components against various CAPRI metrics (i-RMSD, l-RMSD, Fnat, Dock-Q) with a color-coded representation of the clusters.
These are interactive plots. A menu on the top right of the first row (you might have to scroll to the rigth to see it)
allows you to zoom in and out in the plots and turn on and off clusters.

As a reminder, you can also view this report online here
Finally, the report also shows plots of the cluster statistics (distributions of values per cluster ordered according to their HADDOCK rank):

Some single structure analysis
Going back to command line analysis, we are providing in the scripts directory a simple script that extract some model statistics for acceptable or better models from the caprieval steps.
To use is simply call the script with as argument the run directory you want to analyze, e.g.:
./scripts/extract-capri-stats.sh ./runs/run1-CDR-NMR-CSP
View the output of the script expand_more
============================================== == run1/02_caprieval/capri_ss.tsv ============================================== Total number of acceptable or better models: 16 out of 50 Total number of medium or better models: 12 out of 50 Total number of high quality models: 0 out of 50 First acceptable model - rank: 4 i-RMSD: 1.229 Fnat: 0.707 DockQ: 0.744 First medium model - rank: 4 i-RMSD: 1.229 Fnat: 0.707 DockQ: 0.744 Best model - rank: 21 i-RMSD: 1.154 Fnat: 0.828 DockQ: 0.795 ============================================== == run1/05_caprieval/capri_ss.tsv ============================================== Total number of acceptable or better models: 16 out of 50 Total number of medium or better models: 12 out of 50 Total number of high quality models: 2 out of 50 First acceptable model - rank: 1 i-RMSD: 0.778 Fnat: 0.897 DockQ: 0.877 First medium model - rank: 1 i-RMSD: 0.778 Fnat: 0.897 DockQ: 0.877 Best model - rank: 1 i-RMSD: 0.778 Fnat: 0.897 DockQ: 0.877 ============================================== == run1/07_caprieval/capri_ss.tsv ============================================== Total number of acceptable or better models: 16 out of 50 Total number of medium or better models: 12 out of 50 Total number of high quality models: 2 out of 50 First acceptable model - rank: 1 i-RMSD: 1.034 Fnat: 0.879 DockQ: 0.819 First medium model - rank: 1 i-RMSD: 1.034 Fnat: 0.879 DockQ: 0.819 Best model - rank: 3 i-RMSD: 0.822 Fnat: 0.862 DockQ: 0.858 ==============================================
Note that this kind of analysis only makes sense when we know the reference complex and for benchmarking / performance analysis purposes.
Look at the single structure statistics provided by the script
Answer expand_more
In terms of iRMSD values we only observe very small differences in the best model. The fraction of native contacts and the DockQ scores are however improving much more after flexible refinement, but increases again slightly after final minimisation. All this will of course depend on how different are the bound and unbound conformations and the amount of data used to drive the docking process. In general, from our experience, the more and better data at hand, the larger the conformational changes that can be induced.
Is the best model always ranked as first?
Answer expand_more
This is not the case. The scoring function is not perfect, but does a reasonable job in ranking models of acceptable or better quality on top in this case.
Contacts analysis
We have recently added a new contact analysis module to HADDOCK3 that generates for each cluster both a contact matrix of the entire system showing all contacts within a 5Å cutoff and a chord chart representation of intermolecular contacts.
In the current workflow we run, those files can be found in the 11_contactmap directory.
These are again html files with interactive plots (hoover with your mouse over the plots).
This file taken from the pre-computed run can also directly be visualized here
Can you identify which residue(s) make(s) the most intermolecular contacts?
Visualization of the models
To visualize the models from top cluster of your favorite run, start PyMOL and load the cluster representatives you want to view, e.g. this could be the top model from cluster1 for run run1-CDR-NMR-CSP.
These can be found in the runs/run1/09_seletopclusts/ directory
File menu -> Open -> select cluster_1_model_1.pdb
Note that the PDB files are compressed (gzipped) by default at the end of a run. You can uncompress those with the gunzip command. PyMol can directly read the gzipped files.
If you want to get an impression of how well defined a cluster is, repeat this for the best N models you want to view (cluster_1_model_X.pdb).
Also load the reference structure from the pdbs directory, 4G6M-matched.pdb.
Once all files have been loaded, type in the PyMOL command window:
show cartoon util.cbc color yellow, 4G6M_matched
Let us then superimpose all models onto the reference structure:
Let’s now check if the active residues which we have defined (the paratope and epitope) are actually part of the interface. In the PyMOL command window type:
select paratope, (resi 31+32+33+34+35+52+54+55+56+100+101+102+103+104+105+106+151+152+169+170+173+211+212+213+214+216 and chain A) color red, paratope select epitope, (resi 72+73+74+75+81+83+84+89+90+92+94+96+97+98+115+116+117 and chain B) color orange, epitope
Are the residues of the paratope and NMR epitope at the interface?
Note: You can turn on and off a model by clicking on its name in the right panel of the PyMOL window.
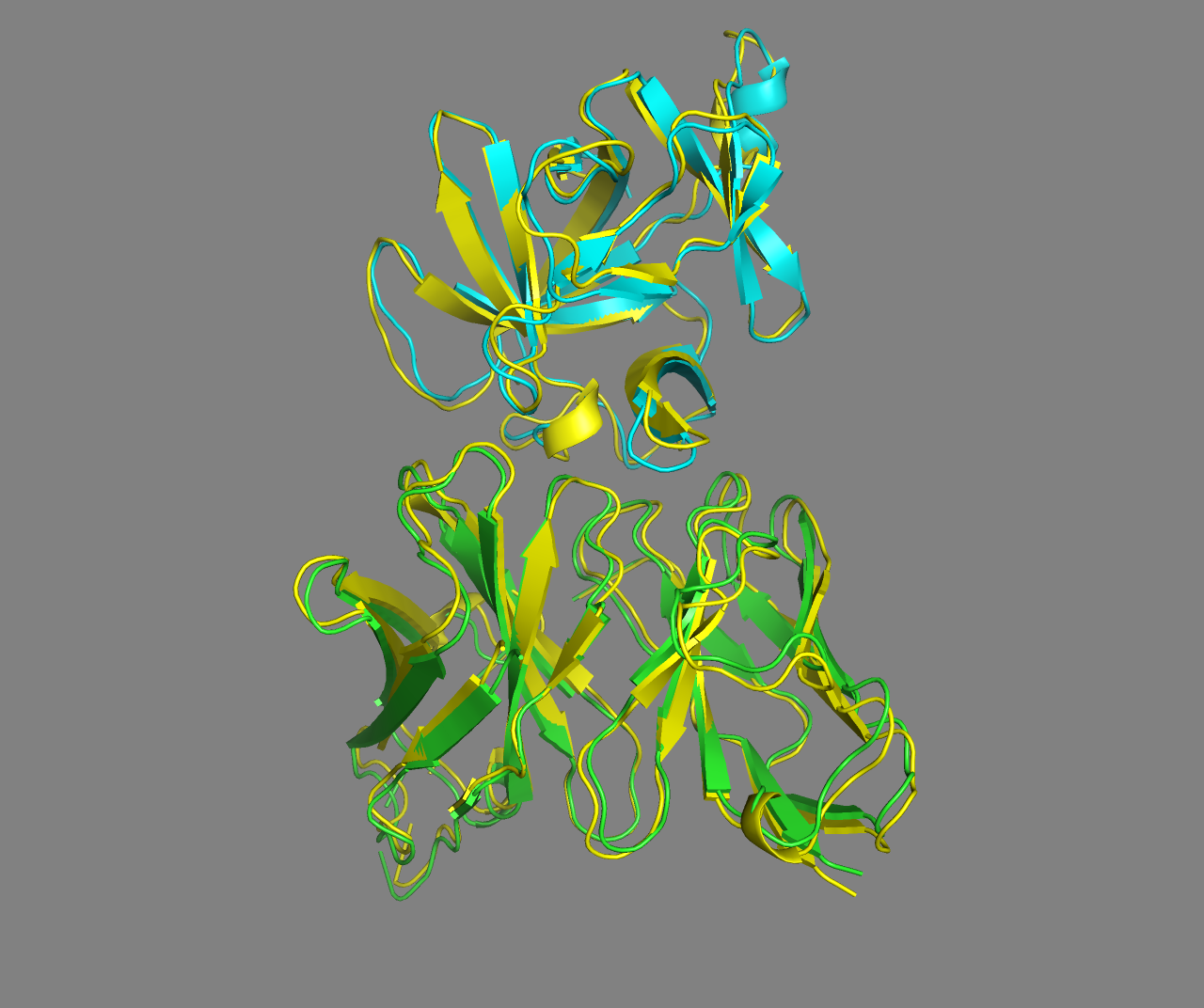
See the overlay of the top ranked model onto the reference structure expand_more
Top-ranked model of the top cluster superimposed onto the reference crystal structure (in yellow)
Conclusions
We have demonstrated the usage of HADDOCK3 in an antibody-antigen docking scenario making use of the paratope information on the antibody side (i.e. no prior experimental information) and a NMR-mapped epitope for the antigen. Compared to the static HADDOCK2.X workflow, the modularity and flexibility of HADDOCK3 allows to customise the docking protocols and to run a deeper analysis of the results. While HADDOCK3 is still very much work in progress, its intrinsic flexibility can be used to improve the performance of antibody-antigen modelling compared to the results we presented in our Structure 2020 article and in the related HADDOCK2.4 tutorial.
BONUS 1: Does the antibody bind to a known interface of interleukin? ARCTIC-3D analysis
Gevokizumab is a highly specific antibody that targets an allosteric site of Interleukin-1β (IL-1β) in PDB file 4G6M, thus reducing its binding affinity for its substrate, interleukin-1 receptor type I (IL-1RI). Canakinumab, another antibody binding to IL-1β, has a different mode of action, as it competes directly with the binding site of IL-1RI (in PDB file 4G6J). For more details please check this article.
We will now use our new software, ARCTIC-3D, to visualize the binding interfaces formed by IL-1β. First, the program retrieves all the existing binding surfaces formed by IL-1β from the PDBe website. Then, these binding surfaces are compared and clustered together if they span a similar region of the selected protein (IL-1β in our case).
We will run an ARCTIC-3D job targeting the uniprot ID of human Interleukin-1 beta, namely P01584.
For this first open the ARCTIC-3D web-server page here.
Insert the selected uniprot ID in the UniprotID field.
Leave the other parameters as they are and click on Submit.
Wait a few seconds for the job to complete or access a precalculated run here.
How many interface clusters were found for this protein?
Once you download the output archive, you can find the clustering information presented in the dendrogram:
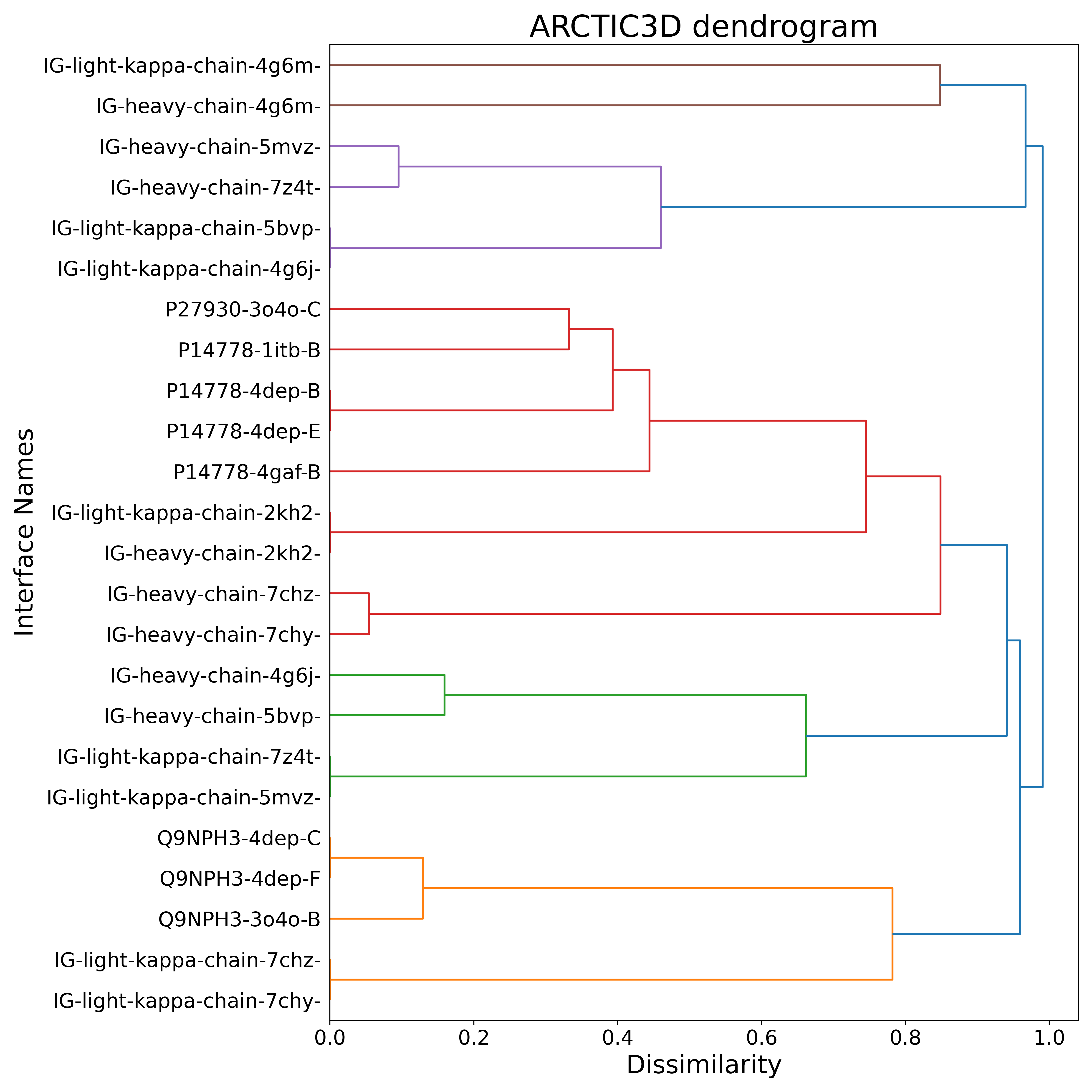
We can see how the two 4G6M antibody chains are recognized as a unique cluster, clearly separated from the other binding surfaces and, in particular, from those proper to IL-1RI (uniprot ID P14778).
Rerun ARCTIC-3D with a clustering threshold equal to 0.95
This means that the clustering will be looser, therefore lumping more dissimilar surfaces into the same object.
You can inspect a precalculated run here.
BONUS 2: How good are AI-based models of antibody for docking?
The release of Alphafold2 in late 2020 has brought structure prediction methods to a new frontier, providing accurate models for the majority of known proteins. This revolution did not spare antibodies, with Alphafold2-multimer and other prediction methods (most notably ABodyBuilder2, from the ImmuneBuilder suite) performing nicely on the variable regions.
For a short introduction to AI and AlphaFold refer to this other tutorial introduction.
CDR loops are clearly the most challenging region to be predicted given their high sequence variability and flexibility. Multiple Sequence Alignment (MSA)-derived information is also less useful in this context.
Here we will see whether the antibody models given by Alphafold2-multimer and ABodyBuilder2 can be used for generating reliabble models of the antibody-antigen complex by docking, instead of the unbound form used in this tutorial, which, in a many cases, will not be available.
Analysing the AI models
We already ran the prediction with these two tools, and you can find the resulting models in the pdbs directory as:
4g6k_Abodybuilder2.pdb4g6k_AF2_multimer.pdb
As was demonstrated in the tutorial, those files must be preprocess for use in HADDOCK. Docking-ready files are also provided in the pdbs directory:
4G6K_abb_clean.pdb4G6K_af2_clean.pdb
Load the experimental unbound structure (4G6K_clean.pdb) and the two AI model in PyMol to see whether they ressemble the experimental unbound structure.
File menu -> Open -> select 4G6K_clean.pdb File menu -> Open -> select 4G6K_abb_clean.pdb File menu -> Open -> select 4G6K_af2_clean.pdb
Align the models to the experimental unbound structure
Which model is the closest to the unbound conformation?
See the RMSD values expand_more
4G6K_abb_clean RMSD = 0.428 Å 4G6K_af2_clean RMSD = 0.765 Å
For docking purposes however, it might be more interesting to know how far are the models from the bound conformation, i.e. the conformation in the antibody-antigen complex. The closer it is the easiest it should become to model the complex by docking. To assess this we can load the structure of the complex in PyMol and align all other structures/models to it.
File menu -> Open -> select 4G6M_matched.pdb
File menu -> Open -> color yellow, 4G6M_matched
Align now the models to the experimental bound structure
alignto 4G6M_matched and chain A
Which model is the closest to the bound conformation?
See the RMSD values expand_more
4G6K_abb_clean RMSD = 0.330 Å 4G6K_af2_clean RMSD = 0.675 Å 4G6K_clean RMSD = 0.393 Å
Docking performance using AI-based antibody models
We can repeat the docking as described above in our tutorial, but using this time either the ABodyBuilder2 or Alphafold2 models as input.
For this modify your haddock3 configuration file, changing the input PDB file of the first molecule (the antibody) using the respective HADDOCK-ready models provided in the pdbs directory.
You will also need to change the restraint filename using to keep the two parts of the antibody together (those files are present in the restraints directory.
Further run haddock3 as described above.
Pre-calculated runs are provided in the runs directory. Analyse your run (or the pre-calculated ones) as described previously.
See the cluster statistics expand_more
============================================== == run1/10_caprieval/capri_clt.tsv ============================================== Total number of acceptable or better clusters: 1 out of 4 Total number of medium or better clusters: 1 out of 4 Total number of high quality clusters: 0 out of 4 First acceptable cluster - rank: 1 i-RMSD: 1.102 Fnat: 0.836 DockQ: 0.805 First medium cluster - rank: 1 i-RMSD: 1.102 Fnat: 0.836 DockQ: 0.805 Best cluster - rank: 1 i-RMSD: 1.102 Fnat: 0.836 DockQ: 0.805 ============================================== == run1-abb/10_caprieval/capri_clt.tsv ============================================== Total number of acceptable or better clusters: 1 out of 5 Total number of medium or better clusters: 1 out of 5 Total number of high quality clusters: 0 out of 5 First acceptable cluster - rank: 1 i-RMSD: 1.103 Fnat: 0.832 DockQ: 0.797 First medium cluster - rank: 1 i-RMSD: 1.103 Fnat: 0.832 DockQ: 0.797 Best cluster - rank: 1 i-RMSD: 1.103 Fnat: 0.832 DockQ: 0.797 ============================================== == run1-af2/10_caprieval/capri_clt.tsv ============================================== Total number of acceptable or better clusters: 2 out of 5 Total number of medium or better clusters: 0 out of 5 Total number of high quality clusters: 0 out of 5 First acceptable cluster - rank: 1 i-RMSD: 2.956 Fnat: 0.375 DockQ: 0.413 Best cluster - rank: 3 i-RMSD: 2.903 Fnat: 0.375 DockQ: 0.327
Which starting structure of the antibody gives the best overall model (irrespective of the ranking)?
Hint: Use the extract-capri-stats.sh script to analyse the various runs and find the best (lowest i-RMSD or highest Dock-Q score).
See single structure statistics expand_more
============================================== == run1/07_caprieval/capri_ss.tsv ============================================== Total number of acceptable or better models: 16 out of 50 Total number of medium or better models: 12 out of 50 Total number of high quality models: 2 out of 50 First acceptable model - rank: 1 i-RMSD: 1.034 Fnat: 0.879 DockQ: 0.819 First medium model - rank: 1 i-RMSD: 1.034 Fnat: 0.879 DockQ: 0.819 Best model - rank: 3 i-RMSD: 0.822 Fnat: 0.862 DockQ: 0.858 ============================================== == run1-abb/07_caprieval/capri_ss.tsv ============================================== Total number of acceptable or better models: 13 out of 50 Total number of medium or better models: 10 out of 50 Total number of high quality models: 2 out of 50 First acceptable model - rank: 1 i-RMSD: 1.249 Fnat: 0.793 DockQ: 0.773 First medium model - rank: 1 i-RMSD: 1.249 Fnat: 0.793 DockQ: 0.773 Best model - rank: 4 i-RMSD: 0.901 Fnat: 0.862 DockQ: 0.857 ============================================== == run1-af2/07_caprieval/capri_ss.tsv ============================================== Total number of acceptable or better models: 23 out of 50 Total number of medium or better models: 1 out of 50 Total number of high quality models: 0 out of 50 First acceptable model - rank: 2 i-RMSD: 2.492 Fnat: 0.466 DockQ: 0.508 First medium model - rank: 22 i-RMSD: 1.780 Fnat: 0.448 DockQ: 0.592 Best model - rank: 22 i-RMSD: 1.780 Fnat: 0.448 DockQ: 0.592
Conclusions - AI-based docking
All three antibody structures used in input give good to reasonable results. The unbound and the ABodyBuilder2 antibodies provided better results, with the best cluster showing models within 1 angstrom of interface-RMSD with respect to the unbound structure. Using the Alphafold2 structure in this case is not the best option, as the input antibody is not perfectly modelled in its H3 loop.
BONUS 3: Ensemble-docking using a combination of experimental and AI-predicted antibody structures
Instead of running haddock3 using a specific input structure of the antibody we can also use an ensemble of all available models.
Such an ensemble can be create from the individual models using pdb_mkensemble from PDB-tools:
pdb_mkensemble 4G6K_clean.pdb 4G6K_abb_clean.pdb 4G6K_af2_clean.pdb >4G6K-ensemble.pdb
This ensemble file is provided in the pdbs directory.
Now we can make use of the flexibility of haddock3 in defining workflows to add a clustering step after the rigid body docking step in order to make sure that models originating from all models will ideally be selected for the refinement steps (provided they do cluster). This modified workflow looks like:
# ====================================================================
# Antibody-antigen docking example with restraints from the antibody
# paratope to the NMR-identified epitope on the antigen
# ====================================================================
# directory in which the scoring will be done
run_dir = "run1-CDR-NMR-CSP"
# compute mode
mode = "local"
ncores=50
# Self contained rundir (to avoid problems with long filename paths)
self_contained = true
molecules = [
"pdbs/4G6K-ensemble.pdb",
"pdbs/4I1B_clean.pdb"
]
# ====================================================================
# Parameters for each stage are defined below, prefer full paths
# ====================================================================
[topoaa]
[rigidbody]
# CDR to NMR epitope ambig restraints
ambig_fname = "restraints/ambig-paratope-NMR-epitope.tbl"
# Restraints to keep the antibody chains together
unambig_fname = "restraints/antibody-unambig.tbl"
sampling = 150
[clustfcc]
[seletopclusts]
top_models = 10
[caprieval]
reference_fname = "pdbs/4G6M_matched.pdb"
[flexref]
tolerance = 5
# CDR to NMR epitope ambig restraints
ambig_fname = "restraints/ambig-paratope-NMR-epitope.tbl"
# Restraints to keep the antibody chains together
unambig_fname = "restraints/antibody-unambig.tbl"
[caprieval]
reference_fname = "pdbs/4G6M_matched.pdb"
[emref]
tolerance = 5
# CDR to NMR epitope ambig restraints
ambig_fname = "restraints/ambig-paratope-NMR-epitope.tbl"
# Restraints to keep the antibody chains together
unambig_fname = "restraints/antibody-unambig.tbl"
[caprieval]
reference_fname = "pdbs/4G6M_matched.pdb"
[clustfcc]
[seletopclusts]
top_models = 4
[caprieval]
reference_fname = "pdbs/4G6M_matched.pdb"
[contactmap]
# ====================================================================Our workflow consists of the following 14 modules:
topoaa: Generates the topologies for the CNS engine and build missing atomsrigidbody: Rigid body energy minimisation - with increased sampling (150 models - 50 per input model)caprieval: Calculates CAPRI metricsclustfcc: Clustering of models based on the fraction of common contacts (FCC)seletopclusts: Selects the top models of all clusters - In this case we select max 10 models per cluster.caprieval: Calculates CAPRI metrics of the selected clustersflexref: Semi-flexible refinement of the interface (it1in haddock2.4)caprievalemref: Final refinement by energy minimisation (itwEM only in haddock2.4)caprievalclustfcc: Clustering of models based on the fraction of common contacts (FCC)seletopclusts: Selects the top models of all clusterscaprievalcontactmap: Contacts matrix and a chordchart of intermolecular contacts
Compared to the original workflow described in this tutorial we have added clustering and cluster selections steps after the rigid body docking.
Run haddock3 with this configuration file as described above.
A pre-calculated run is provided in the runs directory as `run1-ens-clst.
Analyse your run (or the pre-calculated ones) as described previously.
See the cluster statistics expand_more
============================================== == run1-ens-clst//12_caprieval/capri_clt.tsv ============================================== Total number of acceptable or better clusters: 3 out of 7 Total number of medium or better clusters: 1 out of 7 Total number of high quality clusters: 0 out of 7 First acceptable cluster - rank: 1 i-RMSD: 1.276 Fnat: 0.828 DockQ: 0.779 First medium cluster - rank: 1 i-RMSD: 1.276 Fnat: 0.828 DockQ: 0.779 Best cluster - rank: 1 i-RMSD: 1.276 Fnat: 0.828 DockQ: 0.779
See single structure statistics expand_more
============================================== == run1-ens-clst//02_caprieval/capri_ss.tsv ============================================== Total number of acceptable or better models: 50 out of 150 Total number of medium or better models: 25 out of 150 Total number of high quality models: 0 out of 150 First acceptable model - rank: 3 i-RMSD: 1.273 Fnat: 0.672 DockQ: 0.716 First medium model - rank: 3 i-RMSD: 1.273 Fnat: 0.672 DockQ: 0.716 Best model - rank: 46 i-RMSD: 1.154 Fnat: 0.828 DockQ: 0.795 ============================================== == run1-ens-clst//05_caprieval/capri_ss.tsv ============================================== Total number of acceptable or better models: 28 out of 68 Total number of medium or better models: 10 out of 68 Total number of high quality models: 0 out of 68 First acceptable model - rank: 3 i-RMSD: 1.273 Fnat: 0.672 DockQ: 0.716 First medium model - rank: 3 i-RMSD: 1.273 Fnat: 0.672 DockQ: 0.716 Best model - rank: 15 i-RMSD: 1.229 Fnat: 0.707 DockQ: 0.744 ============================================== == run1-ens-clst//07_caprieval/capri_ss.tsv ============================================== Total number of acceptable or better models: 27 out of 68 Total number of medium or better models: 10 out of 68 Total number of high quality models: 3 out of 68 First acceptable model - rank: 1 i-RMSD: 1.454 Fnat: 0.759 DockQ: 0.720 First medium model - rank: 1 i-RMSD: 1.454 Fnat: 0.759 DockQ: 0.720 Best model - rank: 22 i-RMSD: 0.908 Fnat: 0.776 DockQ: 0.803
We started from three different conformations of the antibody: 1) the unbound crystal structure, 2) the ABodyBuilder2 model and 3) the AlphaFold2 model.
BONUS 4: Antibody-antigen complex structure prediction from sequence using AlphaFold2
With the advent of Artificial Intelligence (AI) and AlphaFold we can also try to predict directly the full antibody-antigen complex using AlphaFold. For this we are going to use the AlphaFold2_mmseq2 Jupyter notebook which can be found with other interesting notebooks in Sergey Ovchinnikov ColabFold GitHub repository, making use of the Google Colab CLOUD resources.
Start the AlphaFold2 notebook on Colab by clicking here.
Note: The bottom part of the notebook contains instructions on how to use it.
Setting up the antibody-antigen complex prediction with AlphaFold2
To setup the prediction we need to provide the sequence of the heavy and light chains of the antibody and the sequence of the antigen. These are respectively
- Antibody heavy chain:
QVQLQESGPGLVKPSQTLSLTCSFSGFSLSTSGMGVGWIRQPSGKGLEWLAHIWWDGDES YNPSLKSRLTISKDTSKNQVSLKITSVTAADTAVYFCARNRYDPPWFVDWGQGTLVTVSS
- Antibody light chain:
DIQMTQSTSSLSASVGDRVTITCRASQDISNYLSWYQQKPGKAVKLLIYYTSKLHSGVPS RFSGSGSGTDYTLTISSLQQEDFATYFCLQGKMLPWTFGQGTKLEIK
- Antigen:
VRSLNCTLRDSQQKSLVMSGPYELKALHLQGQDMEQQVVFSMSFVQGEESNDKIPVALGL KEKNLYLSCVLKDDKPTLQLESVDPKNYPKKKMEKRFVFNKIEINNKLEFESAQFPNWYI STSQAENMPVFLGGTKGGQDITDFTMQFVSS
To use AlphaFold2 to predict this antibody-antigen complex follow the following steps:
For your convenience the full sequence with colons is:
QVQLQESGPGLVKPSQTLSLTCSFSGFSLSTSGMGVGWIRQPSGKGLEWLAHIWWDGDESYNPSLKSRLTISKDTSKNQVSLKITSVTAADTAVYFCARNRYDPPWFVDWGQGTLVTVSS:DIQMTQSTSSLSASVGDRVTITCRASQDISNYLSWYQQKPGKAVKLLIYYTSKLHSGVPSRFSGSGSGTDYTLTISSLQQEDFATYFCLQGKMLPWTFGQGTKLEIK:VRSLNCTLRDSQQKSLVMSGPYELKALHLQGQDMEQQVVFSMSFVQGEESNDKIPVALGLKEKNLYLSCVLKDDKPTLQLESVDPKNYPKKKMEKRFVFNKIEINNKLEFESAQFPNWYISTSQAENMPVFLGGTKGGQDITDFTMQFVSS
Define the jobname, e.g. Ab-Ag
In the top section of the Colab, click: Runtime > Run All
(It may give a warning that this is not authored by Google, because it is pulling code from GitHub - you can ignore it).
This will automatically install, configure and run AlphaFold for you - leave this window open. After the prediction complete you will be asked to download a zip-archive with the results (if you configured it to use Google Drive, a result archive will be automatically saved to your Google Drive).
Time to grap a cup of tea or a coffee!
And while waiting try to answer the following questions:
How do you interpret AlphaFold2 predictions? What are the predicted LDDT (pLDDT), PAE, iptm?
Tip: Try to find information about the prediction confidence at https://alphafold.ebi.ac.uk/faq. A nice summary can also be found here.
Pre-calculated AlphFold2 predictions are provided here. This archive contains the five predicted models (the naming indicates the rank), figures (png) files (PAE, pLDDT, coverage) and json files containing the corresponding values (the last part of the json files report the ptm and iptm values).
Analysis of the generated AF2 models
While the notebook is running models will appear first under the Run Prediction section, colored both by chain and by pLDDT.
The best model will then be displayed under the Display 3D structure section. This is an interactive 3D viewer that allows you to rotate the molecule and zoom in or out.
Note that you can change the model displayed with the rank_num option. After changing it execute the cell by clicking on the run cell icon on the left of it.
How similar are the five models generated by AF2?
Consider the pLDDT of the various models (the higher the pLDDT the more reliable the model).
While the pLDDT score is an overall measure, you can also focus on the interface score reported in the iptm score (value between 0 and 1).
See the confidence statistics for the five generated models
Model1: pLDDT=90.4 pTM=0.654 ipTM=0.525
Model2: pLDDT=88.0 pTM=0.65 ipTM=0.522
Model3: pLDDT=88.2 pTM=0.647 ipTM=0.52
Model4: pLDDT=88.0 pTM=0.644 ipTM=0.516
Model5: pLDDT=88.1 pTM=0.641 ipTM=0.512
Note that if you performed a fresh run your results might well differ from those show here.
Based on the iptm scores, would you qualify those models as reliable?
Note that in this case the iptm score reports on all interfaces, i.e. both the interface between the two chains of the antibody, and the antibody-antigen interface
Another useful way of looking at the model accuracy is to check the Predicted Alignment Error plots (PAE) (also referred to as Domain position confidence). The PAE gives a distance error for every pair of residues: It gives the estimate of the position error at residue x when the predicted and true structures are aligned on residue y. Values range from 0 to 35 Angstroms. It is usually shown as a heatmap image with residue numbers running along vertical and horizontal axes and each pixel colored according to the PAE value for the corresponding pair of residues. If the relative position of two domains is confidently predicted then the PAE values will be low (less than 5A - dark blue) for pairs of residues with one residue in each domain. When analysing your complex, the diagonal block will indicate the PAE within each molecule/domain, while the off-diagonal blocks report on the accuracy of the domain-domain placement.
Our antibody-antigen complex consists of three interfaces:
- The interface between the heavy and light chains of the antibody
- The interface between the heavy chain of the antibody and the antigen
- The interface between the light chain of the antibody and the antigen
See the PAE plots for the five generated models
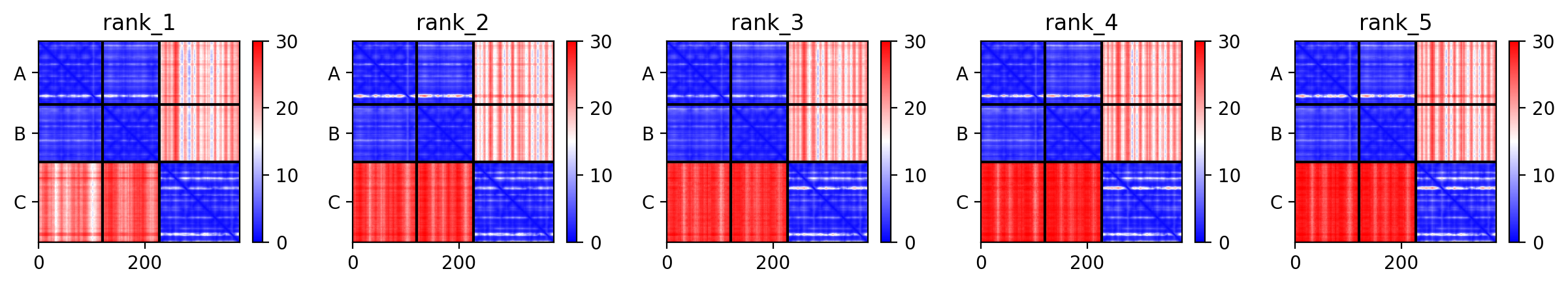
Based on the PAE plots, which interfaces can be considered reliable/unreliable?
Visualization of the generated AF2 models
In order to visualize the models in PyMOL save your predictions to disk or download the precalculated AlphaFold2 model from here.
Start PyMOL and load via the File menu all five AF2 models.
Repeat this for each model (abagtest_2d03e_unrelaxed_rank_X_alphafold2_multimer_v3_model_X_seed_000.pdb or whatever the naming of your model is).
Let us superimpose all models on the antibody (the antibody in the provided AF2 models correspond to chains A and B):
This will align all clusters on the antibody, maximizing the differences in the orientation of the antigen.
Examine the various models. How does the orientation of the antigen differ between them?
Note: You can turn on and off a model by clicking on its name in the right panel of the PyMOL window.
See tips on how to visualize the prediction confidence in PyMOL
When looking at the structures generated by AlphaFold in PyMOL, the pLDDT is encoded as the B-factor.To color the model according to the pLDDT type in PyMOL:
spectrum b **Note** that the scale in the B-factor field is the inverse of the color coding in the PAE plots: i.e. red mean reliable (high pLDDT) and blue unreliable (low pLDDT))
Since we do have NMR chemical shift perturbation data for the antigen, we can check if the perturbed residues are at the interface in the AF2 models. Note that there is a shift in numbering of 2 residues between the AF2 and the HADDOCK models.
Does any model have the NMR-identified epitope at the interface with the antibody?
See the AlphaFold models with the NMR-mapped epitope
It should be clear from the visualization of the NMR-mapped epitope on the AF2 models that none does satisfy the NMR data. To further make that clear we can compare the models to the crystal structure of the complex (4G6M).
For this download and superimpose the models onto the crystal structure in PyMOL with the following commands:
fetch 4G6M
remove resn HOH
color yellow, 4G6M
select 4G6M and chain H+L
alignto sele
See the AlphaFold models superimposed onto the crystal structure of the complex (4G6M)
Congratulations! 🎉
You have completed this tutorial. If you have any questions or suggestions, feel free to contact us via email or asking a question through our support center.
And check also our education web page where you will find more tutorials!